- Home Page
- Company Profile
-
Our Products
- Pharmaceutical Injection
- Adrenaline Injection
- Atropine Sulphate Injection IP
- Atropipe Sulphate Injection
- Adenosine Injection IP
- 5mg Tirofiban Injection
- Methylene Blue Injecton
- Amiodarone Hydrochloride Injection
- Aminophylline Injection IP
- Nutritive Infusion
- Amino Acid Injection With Electrolytes
- Amino Acid Injetion With Electrolytes
- Amino Acids Infusion With Malic Acid For Renal Insufficiency
- Pure Crystalline Amino Acid IV Infusion With Xylitol
- 250mg Amikacin Sulphate Injection
- 500mg Amikacin Sulphate Injection
- Vitamin D3 Injection
- Artestunate Injection
- Artesunate Injection IP
- Ascorbic InjectionIP
- Ascorbic Acid Injection IP
- Azithromycin Injection
- Aztreonam For Injection USP
- Aztreonam For Injection USP
- Frusemide Injection IP
- Betamethasone Sodium Posphate Injection IP
- Pheniramine Maleate Injection IP
- Budesonide Nebuliser Suspension BP
- Hyoscine Butylbromide Injection IP
- Calcium Gluconate And Calcium Lactobionate Injection
- 25000IU Heparin Sodium Injection IP
- 5000IU Heparin Injection IP
- Levocarnitine Injection USP
- Cefepime And Tazobactam For Injection
- Cefepime And Tazobactam For Injection
- Cefepime Injection IP
- Citicoline Injection IP
- Clarithromycin Fon Infusion
- Clindamycin Phosphate Injection IP
- Clindamycin Phosphate Injection IP
- 40mg Enoxaparin Sodium Injection IP
- 60mg Cerebroprotein Hydrolysate Injection
- Colistimethate Sodium Injection
- Colistimethate Sodium Injection
- Colistimethate Sodium Injection
- Colistimethate Sodium Injection
- Acyclovir Intravenous Infusion IP
- Acyclovir Intravenous Infusion IP
- Esomeprazole Sodium Injection
- Methylcobalamin Injection
- Methylcobalamin Nicotinamide And Pyridoxine HCI Injection
- Pantoprazole Injection
- Ceftriaxone Sulbactam Injection
- Ceftriaxone Injection IP
- Teicoplanin Injection IP
- Teicoplanin Injection IP
- Terlipressin Injection
- Thiamine Hydrochloride Injection IP
- Tigecycline Injection IP
- Tranexamic Acid InjctionIP
- Hydrochloride Injection
- Vasopressin Injection IP
- Sodium Valproate Injection
- Vancomycin Hydrochloride Infuion
- 500mg Vancomycin Hydrochloride For Intravenous Infusion IP
- Folic Acid Cyanocobalamin And Niacinamide Injection
- Lignocaine And Adrenaline Injection IP
- 2% Ligncaine Hydrochloride Injection
- 2.5mg Ceftazidime And Avibactam Powder For Concentrate For Solution For Infusion
- 1.5mg Cefoperazone And Sulbactam For Injection
- Pharmaceutical Tablets
- Aceclofenac Paracetamol And Chlorzoxazone Tablets
- Aceclofenac And Paracetamol Tablets
- Aceclofenac Paracetamol ANd Serratiopeptidase Tablets
- Thiocolchicoside And Aceclofenac Tablets
- Calcium And Vitamin D3 TabletsIP
- Cefixime Dispersible Tablets
- Cefixime Dispersible Tablets IP
- Cefixime And Ofloxacin Tablets
- Diclofenac Potassium Paracetamol And Chlorzoxazone Tablets
- Diclofenac Potassium And Paracetamol Tablets
- Diclofenac Sodium Paracetamol And Serratiopeptidase Tablets
- Drotaverine Hydrochloride And Mefenamic Acid Tablets
- Levofloxacin Talets IP
- Metylcobalamin And Gabapentin Tablets
- Levocetirizine Hydrochloride And Montelukast Sodium Tablets
- Ofloxacin Tablets
- Ofloxacin And Ornidazole Tabets
- Cefpodoxime Proxetil Dspersible Talets
- Rabeprazole Sodium Tablets
- Ferrous Ascorbate Folic Acid And Zinc Tablets
- 5mg Levocetirizine Hydrochloride Tablets
- 6mg Deflazacort Tablets
- Amxycillin Potassium Clavulanate And Lactic Acid Bacillus Tablets
- Ondanstron Orally Disingrating Tablets IP
- Azithromycin Tablets IP
- Pharmaceutical Capsules
- Calcium Citrate Maleate Omega-3 Fatty Acid And Vitamin B12 Softgel Capsules
- Calcitriol And Magnesium Softgel Capsules
- Itraconazole Capsules
- 200mg Itraconazole Cpsules
- Methylcobalamin Alpha Lipoic Acid And Folic Acid Capsules
- Pregabalin And Methylcobalamin Capsules IP
- Caronyl Iron Folic Acid Zinc Sulphate And Vitamin B12 Capsules
- Grape Seed Extract And Zinc Sulphate Softgel Capsules
- Omeprazole And Domperidone Capsules IP
- Pantoprazole Sodium And Domeperidone Capsules
- Rabeprazole And Domperidone Capsules
- Rabeprazole And Levosulpiride Capsules
- Ginseng Multivitamin And Multimineral Soft Gel Capsules
- Cholecalciferol 60000IU Softgel Capsules
- Multivitamin Multimineral And Antioxidant Capsules
- Pharmaceutical Syrup
- Aceclofenac And Paracetamo Suspension
- HBR Chlorpheniramine Maleate Syrup
- Zinc Gluconate Syrup
- Disodium Hydrogen Citrate Syrup
- Levocetirizine Dihydrochloride And Montelukast Sodium Suspension
- Ofloxacin Metronidazole And Simethicone Suspension
- Cyropheptadine Hydrocloride And Tricholine Citrate Oral Solution
- Vitamin B-Complex With L-Lysine And Zinc Syrup
- Mefenamic Acid And Paracetamol Suspension
- Paracetamol Phenylephrine Suspension
- Ambroxol HCL Terbutaline Sulphate Guaiphenesin And Menthol Syrup
- Silymarin And D-Panthenol Syrup
- Carbonyl Zinc Sulphate Vitamin B12 And Folic Acid Syrup
- Vitamin B-Complex With L-Lysine And Zinc Syrup
- Pharmaceutical Ointment
- Pharmaceutical Drops
- Energy Drink
- Ayurvedic Herbal Medicines
- Gastorun Herbal Antacid Capsules
- Honeymik-TS Ayurvedic Cough Syrup
- Giloy Panch Syrup
- Immune Amrit Syrup
- Livorum Ayurvedic Liver Syrup
- Mikrrhoid Piles Capsules
- Mikrroid Piles Syrup
- Neem Karela Jamun Juice
- Platomik-U Health Tonic
- Platomik-UP_Carica Papaya Leaf Extract Giloy With Goat Milk Capsules
- Rakht-Mik Blood Purifier Capsules
- Rakht-Mik Blood Purifier Syrup
- Rumstone Capsules
- RumstoneUrinary Stoe Herbal Tonic
- Samaran Pushpi Herbal Brain Booster Tonic
- Triphala Juice
- Vomik Herbal Pain Relief Oil
- Wheat Grass Juice
- Herbal Digestive Tonic
- Pharmaceutical Injection
- Contact Us
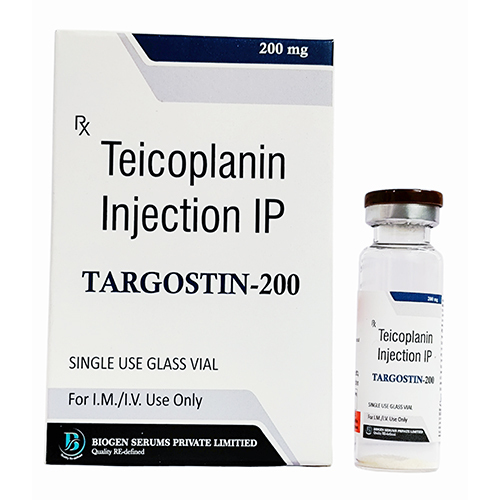
Teicoplanin Injection IP
Product Details:
- Origin of Medicine India
- Pacakaging (Quantity Per Box) 1 vial + 1 ampoule of sterile water
- Life Span 24 months from the date of manufacture
- Brand Name Teicoplanin Injection IP
- Indication Serious Gram-positive infections including MRSA, bone and joint infections, endocarditis, septicemia
- Salt Composition Teicoplanin
- Packaging Type Vial with sterile water ampoule
- Click to view more
X
Teicoplanin Injection IP Price And Quantity
- 100 Pack
- Schedule H Prescription Drug
- Clear, colorless to pale yellow
- 200 mg or 400 mg per vial (as per pack)
- Possible rash, pruritus, fever, and rarely nephrotoxicity or ototoxicity
- As per batch/manufacturer
- Known hypersensitivity to teicoplanin or other glycopeptides
- Intravenous (IV) or Intramuscular (IM)
- 7.0 to 8.5
- Monitor renal function during prolonged therapy
Teicoplanin Injection IP Product Specifications
- Teicoplanin Injection IP
- As directed by physician, typically 612 mg/kg body weight
- 24 months from the date of manufacture
- 1 vial + 1 ampoule of sterile water
- Lyophilized powder for injection
- India
- Prescription only
- Adults and children with Gram-positive bacterial infections
- Antibacterial (glycopeptide antibiotic)
- Reconstitute with sterile water for injection before use. Use immediately after reconstitution.
- One vial
- Vial with sterile water ampoule
- Teicoplanin
- Suitable for all age groups under medical supervision
- Injection
- Teicoplanin
- Serious Gram-positive infections including MRSA, bone and joint infections, endocarditis, septicemia
- Store below 25C. Protect from light and moisture.
- Schedule H Prescription Drug
- Clear, colorless to pale yellow
- 200 mg or 400 mg per vial (as per pack)
- Possible rash, pruritus, fever, and rarely nephrotoxicity or ototoxicity
- As per batch/manufacturer
- Known hypersensitivity to teicoplanin or other glycopeptides
- Intravenous (IV) or Intramuscular (IM)
- 7.0 to 8.5
- Monitor renal function during prolonged therapy
Teicoplanin Injection IP Trade Information
- Cash in Advance (CID)
- 5000 Pack Per Month
- 10 Days
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- All India
Product Description
Experience the exclusive power of Teicoplanin Injection IP, meticulously formulated in select strengths of 200 mg or 400 mg per vial. This antibacterial is unassailable against serious Gram-positive infections, including MRSA, delivering brilliant efficacy where it matters most. Each vial comes with sterile water for injection, ensuring clear, colorless to pale yellow solutions upon reconstitution. Enjoy peace of mind with the finest manufacturing standards from India-a prescription-only solution that combines advanced safety with discounted access through select suppliers. Store below 25C, protected from light and moisture.
Versatility and Application of Teicoplanin Injection IP
Teicoplanin Injection IP is suitable for all age groups, making it an adaptable solution for both adults and children with Gram-positive bacterial infections. Its surface of application is strictly parenteral, administered via intravenous (IV) or intramuscular (IM) routes. The site of application is the bloodstream, ensuring rapid delivery to systemic infection sites. For optimal results, administration should always be under medical supervision, maximizing both safety and therapeutic effectiveness.
Global Export Markets and Assurance in Supply Chain
Teicoplanin Injection IP is dispatched to main export markets including Asia, Africa, and the Middle East. Each unit undergoes rigorous packing & dispatch procedures, ensuring stability and potency upon arrival. Logistics are managed efficiently to expedite delivery times for sample requests as well as bulk orders. The product is available with necessary certifications, maintaining compliance with international standards for quality and safety. Sample availability further demonstrates commitment to excellence in supply partnerships.
Versatility and Application of Teicoplanin Injection IP
Teicoplanin Injection IP is suitable for all age groups, making it an adaptable solution for both adults and children with Gram-positive bacterial infections. Its surface of application is strictly parenteral, administered via intravenous (IV) or intramuscular (IM) routes. The site of application is the bloodstream, ensuring rapid delivery to systemic infection sites. For optimal results, administration should always be under medical supervision, maximizing both safety and therapeutic effectiveness.
Global Export Markets and Assurance in Supply Chain
Teicoplanin Injection IP is dispatched to main export markets including Asia, Africa, and the Middle East. Each unit undergoes rigorous packing & dispatch procedures, ensuring stability and potency upon arrival. Logistics are managed efficiently to expedite delivery times for sample requests as well as bulk orders. The product is available with necessary certifications, maintaining compliance with international standards for quality and safety. Sample availability further demonstrates commitment to excellence in supply partnerships.
FAQ's of Teicoplanin Injection IP:
Q: How should Teicoplanin Injection IP be administered?
A: Teicoplanin Injection IP is reconstituted with sterile water for injection and administered either intravenously (IV) or intramuscularly (IM). Follow the dosage guidelines provided by the prescribing physician.Q: What are the main indications for using Teicoplanin Injection IP?
A: This injection is indicated for serious Gram-positive bacterial infections, including MRSA, bone and joint infections, endocarditis, and septicemia.Q: When should I avoid using Teicoplanin Injection IP?
A: Avoid this medication in cases of known hypersensitivity to teicoplanin or other glycopeptides. If you experience severe allergic reactions or side effects, consult your healthcare provider immediately.Q: Where is Teicoplanin Injection IP manufactured and exported from?
A: This medicine originates in India and is exported to various international markets, including Asia and Africa, ensuring global access to quality antibacterial treatment.Q: What process should be followed after reconstituting Teicoplanin Injection IP?
A: After using the provided sterile water to reconstitute the vial, ensure the solution is clear and colorless to pale yellow. Use the dose immediately as directed by your physician.Q: What precautions need to be considered during prolonged therapy?
A: During extended treatment with Teicoplanin Injection IP, regular monitoring of renal function is recommended to prevent potential nephrotoxicity.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email




 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese